Macular Degeneration Research
Stem Cells to Genes
Macular degeneration research involves many aspects of this disease including prevention, treatment, nutrition, and ways to better utilize the vision one does have.
AMD Clinical Trials
Information on macular degeneration clinical trials are available at the National Institutes of Health website. It lists not only studies in the U.S. but internationally as well.
There are different phases of studies as well as strict criteria to be eligible to be in a macular degeneration study.
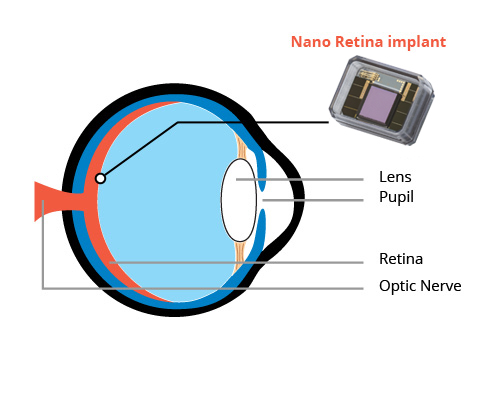
Retina Implant
Retinal implants used advanced technology for individuals with macular degeneration, a condition that that leads to loss of central vision. These implants aim to restore vision by bypassing damaged cells in the retina and directly stimulating the remaining healthy cells or the optic nerve.
Some implants involves a small electronic implant that is surgically placed on the surface of the retina. It works in conjunction with a camera mounted on glasses worn by the user. The camera captures images, which are then processed and wirelessly transmitted to the implant. The implant stimulates the remaining functional cells in the retina, allowing the user to perceive patterns of light and regain some visual function.
While retinal implants can provide some benefits to some individuals with macular degeneration, they are not a cure for the condition and do not fully restore normal vision. Additionally, the effectiveness of these implants vary among individuals, and they may not be suitable for everyone with macular degeneration. As with any surgical procedure, there are risks involved, and individuals considering retinal implants should consult with their healthcare provider to determine if they are appropriate candidates.
Research and development in the field of retinal implants continue to advance, with ongoing efforts to improve the technology and expand its applicability to a broader range of patients with vision loss.
Macular Pigment Research Center
A research center that focuses soley on the carotenoids that benefit the eye, lutein, zeaxanthin and meso-zeaxanthin, is proving in clinical trials that the health of the macula pigment does indeed affect one's vision.
Known as the Macular Pigment Research Center located in Waterford, Ireland, a staff of scientists devote their attention to understanding the role of nutrition in building the macular pigment.
Patients from the center share their success stories in a recent documentary. Go here to watch it on-line:
Wet Macular Degeneration Research
Macular degeneration clinical trials for wet macular degeneration treatment involve many different treatment options.
There are studies looking at new combinations of existing treatment - such as anti-VEGF injections and photodynamic therapy.
Other studies are looking at radiation therapy in conjunction with macular degeneration medication or injections. To see what's available for wet macular degeneration research click here:
Wet Macular Degeneration Clinical Trials
Dry Macular Degeneration Research
Dry AMD is the most common type of macular degeneration - up to 80-85% of patients have this form of macular degeneration.
Dry macular degeneration clinical trials are becoming more available as new treatments and earlier intervention is being researched. From nanosecond laser therapy of macular drusen to eye injections, these clinical trials treat patients that have intermediate to advanced dry AMD. Therapies are investigating ways to prevent the progression of dry AMD into the more advanced geographic atrophy or from turning into wet AMD.
There are several things you will want to know when considering a clinical trial.
1. Where is the Study Being Conducted
The clinical trials that are listed at www.clinicaltrials.gov include studies that are offered in the United States as well as abroad. So it is important to look at the location of the study. Often the study will have several study locations.
2. What Phase Is the Study In
There are four phases of clinical trials. Phase 1 is the first stage that focuses on safety and recruits the smallest number of participants, while a Phase 2 is looking more at the effectiveness of the therapy.
A Phase 3 will recruit a larger population and will continue to look at safety and effectiveness and may also evaluate different therapeutic dosages.
Dry Macular Degeneration Clinical Trials
Gene Therapy for Macular Degeneration
Gene therapy to halt or slow the progression of macular degeneration is in research at several different eye centers.
Research using gene therapy to treat neovascular or wet macular degeneration works by delivering a gene that inhibits blood vessel growth that leads to leaking blood vessels under the retina which distorts one's vision leading to several different types of vision changes including the loss of one's central or straight ahead vision.
Gene Therapy for Macular Degeneration
Dry Macular Degeneration Treatment Breakthrough
The Bionic Vision Restoration System, by Pixium Vision received approval in the fall of 2017 from French regulators for the implant of a retinal chip in patients who have advanced dry macular degeneration (AMD).
The chip is surgically placed in the subretinal space and works in conjunction with a headworn camera and a small data transmitter.
Artificial stimulation of degenerated photoreceptor cells allows for better communication between the eye and the brain.
Dry Macular Degeneration Treatment Breakthrough with Pixium Vision
Research for both dry and wet forms of macular degeneration is progressing as ways to intervene earlier in the disease is helping to prevent vision loss and/or restore some central vision loss.
Go from Macular Degeneration Research to WebRN Macular Degeneration Home
√ Prevention of Macular Degeneration?
√ Tips for Daily Living?
√ Food Suggestions for a Macular Degeneration Diet?
√ Ideas on Visual Aids to Maximize your Sight?
If you said "yes" to any of the above, sign up for the monthly Macular Degeneration News.